March 27, 2021 – What’s a More Useful Measurement – A Particle’s Hydrodynamic Diameter or its ‘Core’ Diameter?
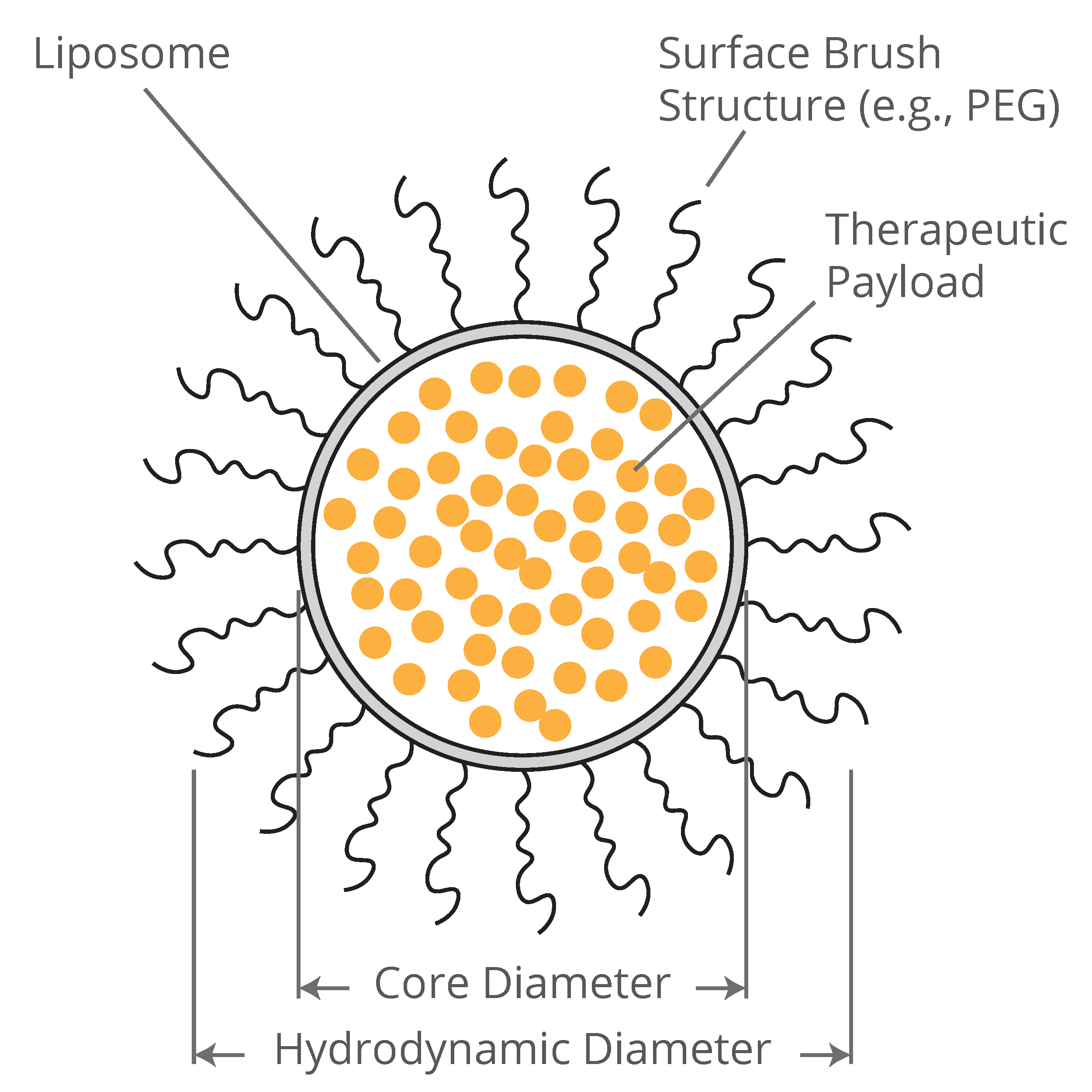
A sketch of a liposome with a surface brush structure, providing a good illustration of the difference between hydrodynamic and core diameters
The short answer: They are just measurements of different properties of the same particle, and the value of each depends on the application. Let’s say you want to know how fast a particle will diffuse in some aqueous solution: Then you want the hydrodynamic diameter. Let’s say you want to know how much of a drug formulation you can put inside a nanoparticle: Then you want the core diameter. The hydrodynamic diameter of a particle is essentially the diameter of a hard sphere that would experience the same drag force as the particle in question when moving in a liquid. Put another way, it is the diameter of a hard sphere that diffuses at the same rate as the particle in question.
Commonly used nanoparticle analysis techniques, dynamic light scattering (DLS) and nanoparticle tracking analysis (NTA), quantify the diffusion behavior of particles in a sample and return a calculation of their hydrodynamic diameters. Calculating hydrodynamic diameter from a diffusion measurement involves significant assumptions: In particular, the calculation is sensitive to the viscosity of the suspending medium, which can be difficult to measure and itself depends strongly on temperature. More importantly, even if the assumptions are accurate the result of this calculation is the hydrodynamic diameter, which may be a useful parameter for certain applications (e.g., diffusive transport experiments) but which does not necessarily say much about the actual diameter of the particle.
Here is a real example: The circulation lifetime of lipid nanoparticle (LNP) and liposome-based drug delivery vehicles in nanomedicine applications is commonly extended by incorporating a “brush” structure of long polyethylene glycol (PEG) chains to the surface of the particles. The PEG brush structure helps the particles avoid clearance from circulation by the body’s liver or kidneys, prolonging the drug’s active lifetime and ultimately increasing the drug’s efficacy. The RNA SARS-CoV-2 vaccines by Moderna and Pfizer-BioNtek are structured in this way. For a particle of this type, the hydrodynamic radius may be significantly larger than the particle’s core size, since the PEG brush structure increases drag on the particle and gives it the diffusion properties of a hard sphere of a larger diameter.
Who cares? Well, the drug maker does, because an important metric for dosing any particle-based drug is the payload capacity of the particles: The volume of each particle available to be loaded with active pharmaceutical ingredients (API). The payload capacity of a particle is much more directly measured by the particles’ core diameter, since the payload is contained inside the particle’s core, and not in its brush surface coating. Using the hydrodynamic diameter of a particle to calculate the payload volume of a PEGylated particle can lead to significant errors: For a 100 nm diameter particle with a 15 nm-long PEG brush on its surface (yielding an approximately 130 nm hydrodynamic diameter), the calculated payload capacity might be as much as 220% of actual!
So how to measure the core diameter? Cryo-electron microscopy is a good choice. Don’t have that kind of time or money? Try Spectradyne’s nCS1, which uses microfluidic resistive pulse sensing (MRPS) to directly measure the core diameter of particles (and their concentration, another key property for dosing nanomedicines) quickly and accurately. Learn more by sending us a sample for a free measurement.
Please continue to follow our blog as we share insights, technical details, and generally geek-out with you about nanoparticle science!
Email us for more information, or to discuss your particular application directly.