

We are grateful for the enormous efforts of scientists, production experts and logistics teams that have made possible the deployment of COVID vaccines to our communities, families and employees. From a science perspective, the messenger-RNA vaccine technology developed for some of these products is incredibly powerful, and hopefully a gamechanger for vaccine development strategies in the future.
Amidst this unprecedented global vaccination effort, do you ever wonder what particles could be in the COVID vaccine you may have received? We know the vaccines from Pfizer-BioNTech and Moderna are based on lipid nanoparticles (LNPs) loaded with messenger-RNA — but what does the size distribution of these particles look like?
We found out! Spectradyne obtained a small residual sample of one of the mainstream two-dose vaccines and analyzed it using Spectradyne's nCS1TM. Since only 3 μL of the sample are required for measurement, we can be confident that no person was prevented from receiving a dose as a result of this testing!
The measurement results are shown in the figure. As expected for a LNP formulation, the particle size distribution is overwhelmingly dominated by particles smaller than 100 nm in diameter. Notably, the sample includes a readily measured concentration of particles as large as 2 μm in diameter.
While the presence of micron-sized particles in the sample comes as a surprise, these particles are likely degradation products in the sample (e.g., aggregates of smaller particles) that formed over time before measurement: While an effort was made to maintain 4°C storage conditions for the vaccine, the measurement of the sample was performed over 24 hours after its recommended expiration time for delivery to a recipient. As we've shown before in protein-based drug formulations, degradation products increase steadily over time as the formulation experiences stress (thermal stress or other). It certainly would be interesting to monitor the concentration of large particles forming in the COVID vaccine over time and validate the recommended expiration time of the preparations... another time!
From sunscreen to liquor to the COVID vaccines, nanoparticles are all around us — let's keep measuring!
Please continue to follow our blog as we share insights, technical details, and generally geek-out with you about nanoparticle science!
Email us for more information, or to discuss your particular application directly.
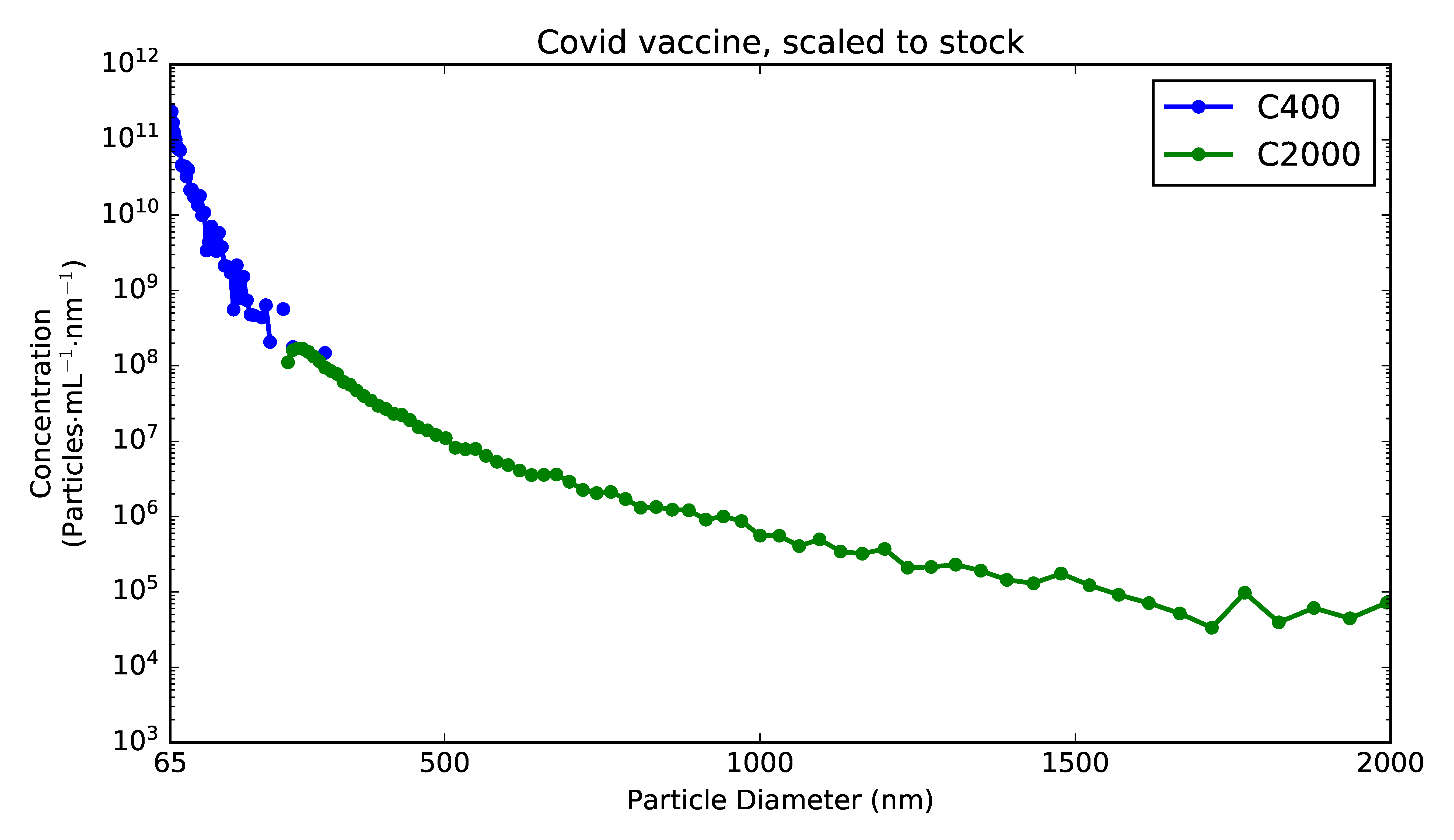
Figure 1. Measurement of a commercial two-part COVID vaccine, plotting concentration spectral density (CSD) versus particle diameter, using two microfluidic cartridges to cover the full size range.